Armata Pharmaceuticals reported Q4 2025 grant revenue of $1.09M, beating the $0.74M analyst estimate, but EPS of -$3.42 missed the consensus of -$0.19 by a wide margin. Full-year net loss widened sharply to $173.8M. Shares traded up roughly 4.87% to $8.61 on earnings day, reflecting cautious optimism over pipeline progress.
About Armata Pharmaceuticals
Armata Pharmaceuticals, Inc.(NYSE American: ARMP) is a late clinical-stage biotechnology company headquartered in Los Angeles, California, founded in 1989. The company focuses on the development of high-purity, pathogen-specific bacteriophage (phage) therapeutics for the treatment of antibiotic-resistant and difficult-to-treat bacterial infections.
Its proprietary phage-based technology platform is used to build both natural and synthetic phage candidate pipelines targeting pathogens such as Pseudomonas aeruginosa, Staphylococcus aureus, and others. Lead programs include AP-SA02 for complicated S. aureus bacteremia and AP-PA02 for chronic pulmonary Pseudomonas aeruginosa infections. The company also holds a research collaboration with Merck Sharp and Dohme Corp. for synthetic phage candidates.
As of March 2026, Armata has a market capitalization of approximately $313.5 million, approximately 60 employees, and no dividend yield. The company carries a negative P/E ratio given its pre-revenue, clinical-stage status. Total revenue for trailing twelve months stands at approximately $5.05M.
Top Financial Highlights
- Q4 2025 Grant Revenue was $1.1 million, a slight decline from $1.2 million in Q4 2024, reflecting MTEC’s contributions to the AP-SA02 program
- Full-Year 2025 Grant Revenue was $4.904 million, down from $5.174 million in 2024
- Q4 2025 Net Loss was $124.3 million, or $(3.42) per share (basic and diluted), versus net income of $2.6 million or $0.07 per share in Q4 2024
- Full-Year 2025 Net Loss totaled $173.8 million, a dramatic increase from the $18.9 million net loss in FY2024
- Full-Year 2025 EPS (basic) was $(4.80), versus $(0.52) in FY2024
- Total Operating Expenses for FY2025 were $41.54 million, down from $47.61 million in FY2024
- R&D Expenses declined to $23.72 million in FY2025, from $34.43 million in FY2024, driven by reduced clinical trial activity
- G&A Expenses were $12.41 million for FY2025, versus $13.18 million in FY2024
- Impairment Charge of $5.412 million was recognized in Q4 2025, related to vacated office and R&D space in Marina del Rey, California, with no such charge in 2024
- Operating Loss for Q4 2025 was $13.8 million, compared to $10.5 million in Q4 2024
- Non-Cash Convertible Loan Fair Value Loss was $120.96 million in FY2025, compared to a $31.40 million gain in FY2024, the primary driver of the net loss widening
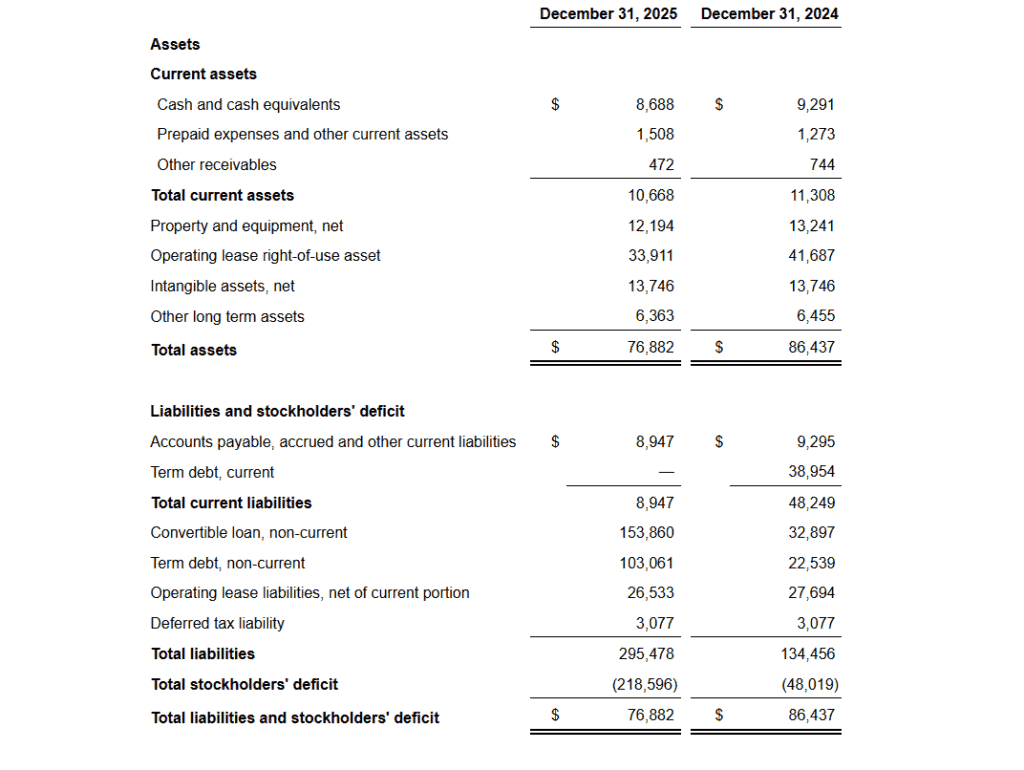
- Cash and Equivalents (including restricted cash) stood at $14.1 million as of December 31, 2025, compared to $14.8 million at year-end 2024
- Net Cash Used in Operating Activities was $25.76 million in FY2025, improving from $37.55 million in FY2024
- Total Liabilities rose to $295.5 million, resulting in a stockholders’ deficit of $218.6 million
- Approximately 36.6 million common shares were outstanding as of March 18, 2026
Beat or Miss?
| Metric | Reported | Estimated | Difference / Analysis |
| Q4 2025 EPS | ($3.42) | ($0.19) | Miss by $3.23 – primarily driven by a $105.8M non-cash convertible loan fair value loss |
| Q4 2025 Revenue | $1.09M | $0.74M | Beat by ~47% – grant revenue from MTEC for AP-SA02 exceeded expectations |
| Full-Year EPS (Basic) | ($4.80) | N/A | No full-year consensus; large non-cash charges dominated |
| Full-Year Revenue | $4.904M | N/A | Revenue missed analyst-estimated trajectory; down 5.2% YoY |
| Operating Cash Burn | -$25.76M | N/A | Improved from -$37.55M in FY2024 – a positive operational trend |
What Leadership Is Saying?
“Compelling efficacy data from the Phase 1b/2a randomized controlled study of intravenously administered AP-SA02, including the favorable safety and tolerability profile, underscore the precision infection control enabled by Armata’s well-characterized, high-purity phage cocktails, and provide strong rationale for advancement of AP-SA02 into late-stage clinical development.
Subject to review and feedback from the U.S. Food and Drug Administration, we are committed to developing a superiority pivotal trial with the goal of introducing AP-SA02 as a new standard of care for complicated Staphylococcus aureus bacteremia, a common, extremely severe, and often deadly infection.” – Dr. Deborah Birx, Chief Executive Officer, Armata Pharmaceuticals
“We have made tremendous progress this year advancing phage-based therapeutics as potential treatments for both acute and chronic bacterial infections, and with line-of-site to the potential initiation of a Phase 3 study in 2026, we believe we are well positioned to bring new hope to patients with significant unmet medical need, while creating long-term value for our shareholders.” – Dr. Deborah Birx, Chief Executive Officer, Armata Pharmaceuticals
Historical Performance
Full-Year Comparison FY2025 vs FY2024
G&A Expenses$12.409M$13.184M-5.90%Operating Loss-$36.634M-$42.436MImproved by 13.7%Net Loss-$173.799M-$18.916M-819% (driven by non-cash items)EPS (Basic)($4.80)($0.52)Wider loss per shareNet Cash Used in Operations-$25.763M-$37.551MImproved by 31.4%Cash and Equivalents (incl. restricted)$14.078M$14.771M-4.70%
| Category | FY2025 | FY2024 | Change (%) |
| Grant and Award Revenue | $4.904M | $5.174M | -5.20% |
| Total Operating Expenses | $41.538M | $47.610M | -12.70% |
| R&D Expenses | $23.717M | $34.426M | -31.10% |
| Total Liabilities | $295.478M | $134.456M | 119.80% |
| Stockholders’ Deficit | -$218.596M | -$48.019M | Widened significantly |
Quarterly Comparison Q4 2025 vs Q4 2024
| Category | Q4 2025 | Q4 2024 | Change (%) |
| Grant Revenue | $1.1M | $1.2M | -8.30% |
| R&D Expenses | $6.1M | $8.5M | -28.20% |
| G&A Expenses | $3.4M | $3.3M | 3.00% |
| Impairment Expense | $5.4M | $0 | New charge in Q4 2025 |
| Operating Loss | -$13.8M | -$10.5M | -31.40% |
| Net Income (Loss) | -$124.3M | +$2.6M | Swung to deep loss |
| EPS (Basic) | ($3.42) | $0.07 | Reversal from gain to loss |
Competitor Comparison
Armata operates in a niche bacteriophage therapeutics space. Direct public competitors with comparable phage programs are limited. The closest biotech peers in antibiotic-resistant infection therapeutics used for context include Destiny Pharma (DEST.L), Lytone Pharma, and SIGA Technologies. Specific full-year 2025 financials for pure-play phage competitors are not publicly available; the table below uses known comparable biotech peers in the antibiotic-resistant infection space for general context.
| Company | Ticker | Focus | FY2025 Revenue (est.) | FY2024 Net Loss | YoY Change |
| Armata Pharmaceuticals | ARMP | Bacteriophage (phage) therapy | $4.9M (grant only) | -$18.9M (FY2024) | Loss widened to -$173.8M (non-cash driven) |
| SIGA Technologies | SIGA | Antiviral/antibiotic-resistant | Primarily commercial revenues | Positive net income | N/A |
| Iterion Therapeutics | ITRN | Clinical-stage biopharma | Minimal/grant revenue | Deep net losses | N/A |
How the Market Reacted?
ARMP shares traded up approximately 4.87%, reaching $8.61 during midday trading on March 25, 2026, the day the earnings report was released. This positive movement came despite the headline EPS miss of $3.23, suggesting investors focused on the revenue beat, the AP-SA02 pipeline progress, and the FDA’s Qualified Infectious Disease Product (QIDP) designation for AP-SA02, which extends market exclusivity incentives.
The company’s stock had already been under pressure heading into earnings, declining nearly 20% in the week prior to the March 19 reporting delay announcement, and had traded as low as $8.54. Analyst sentiment remains broadly constructive, with HC Wainwright raising its price target to $15.00 and JonesTrading initiating coverage with a Buy rating, both citing the late-stage pipeline catalysts as the key value driver. The stock’s 52-week range of $0.90 to $16.34 reflects the significant volatility tied to clinical and regulatory milestone events.



