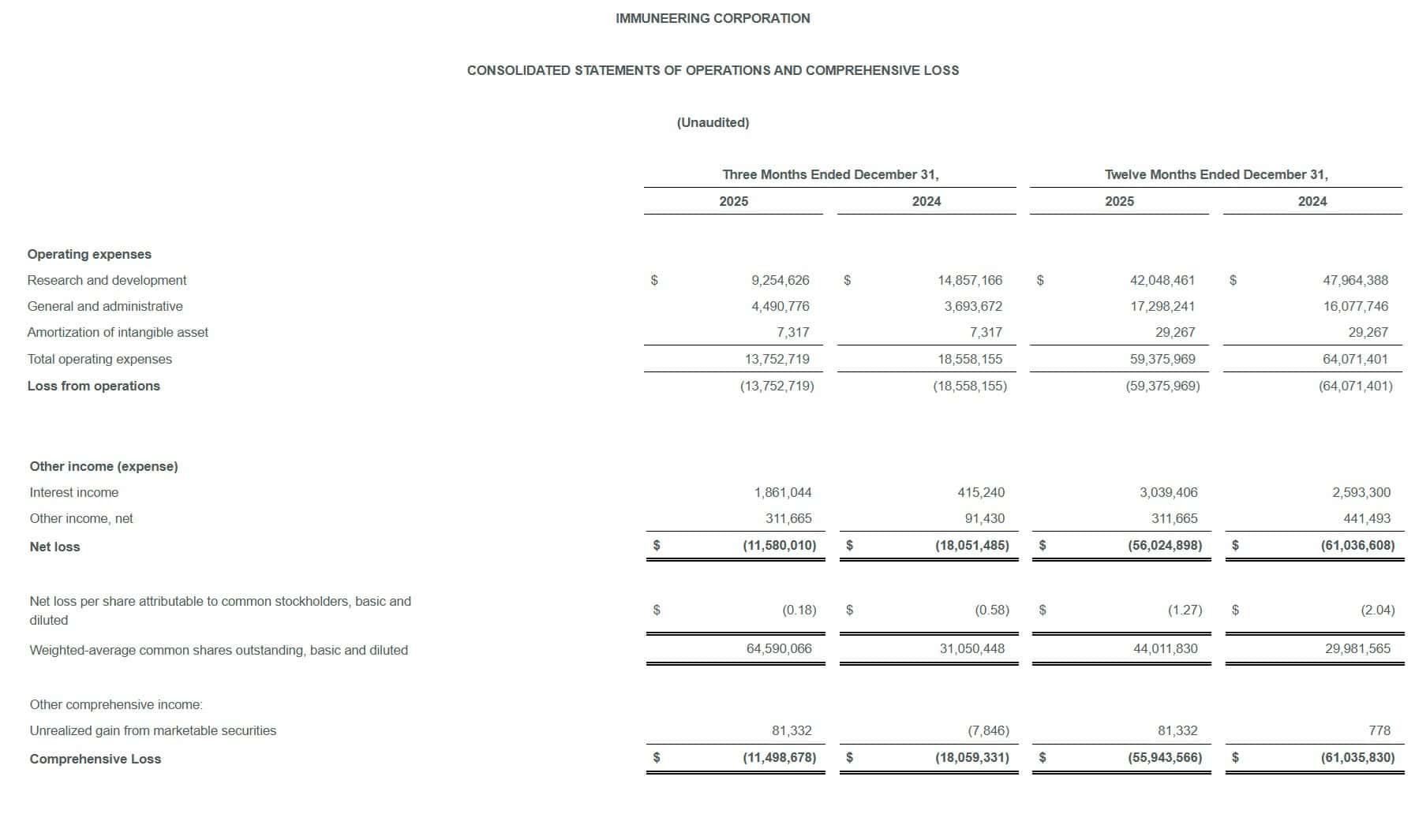
Immuneering (IMRX) reported a Q4 2025 net loss of -$0.18 per share, significantly beating the Wall Street consensus estimate of -$0.31 per share by approximately 42%. The company generated no revenue as a clinical-stage biotech, but narrowed its net loss to $11.6 million versus $18.1 million a year ago. Shares closed at $5.33 on March 6, 2026, up 7.03% on the day, before slipping in after-hours trading. Cash reserves surged to $217 million, providing runway into 2029.
About Immuneering Corporation
Immuneering Corporation (Nasdaq: IMRX) is a late-stage clinical oncology company focused on developing a new category of cancer medicines called Deep Cyclic Inhibitors (DCIs), designed to improve overall survival by shrinking tumors durably, preserving body mass, and minimizing side effects. Founded in 2008 and originally headquartered in Cambridge, Massachusetts, the company now operates from New York, NY. Immuneering went public on July 30, 2021, and as of early March 2026 carries a market capitalization of approximately $305–$322 million.
The company’s lead product candidate, atebimetinib, is an oral, once-daily Deep Cyclic Inhibitor of MEK being evaluated in first-line pancreatic cancer and solid tumors harboring RAS/RAF mutations. Immuneering employs approximately 54 people and currently generates no product revenue, as it remains in the clinical development stage. The stock trades at a price-to-earnings ratio of approximately -2.65 and has a 52-week trading range of $1.10–$10.08. In December 2025, Immuneering was added to the Nasdaq Biotechnology Index (NBI).
Top Financial Highlights
- Total Revenue: $0 — Immuneering is a pre-revenue, clinical-stage company with no product sales.
- Q4 2025 Net Loss: $11.6 million (or -$0.18/share), a significant improvement from $18.1 million (or -$0.58/share) in Q4 2024.
- Full Year 2025 Net Loss: $56.0 million (or -$1.27/share), down from $61.0 million (or -$2.04/share) in FY2024.
- Q4 2025 R&D Expenses: $9.3 million, down 37.7% from $14.9 million in Q4 2024.
- Full Year 2025 R&D Expenses: $42.0 million, down from $48.0 million in FY2024.
- Q4 2025 G&A Expenses: $4.5 million, up from $3.7 million in Q4 2024.
- Full Year 2025 G&A Expenses: $17.3 million, up from $16.1 million in FY2024.
- Total Q4 2025 Operating Expenses: $13.75 million, down from $18.56 million in Q4 2024.
- Interest Income (Q4 2025): $1.86 million, up from $0.42 million in Q4 2024, reflecting a much larger cash balance.
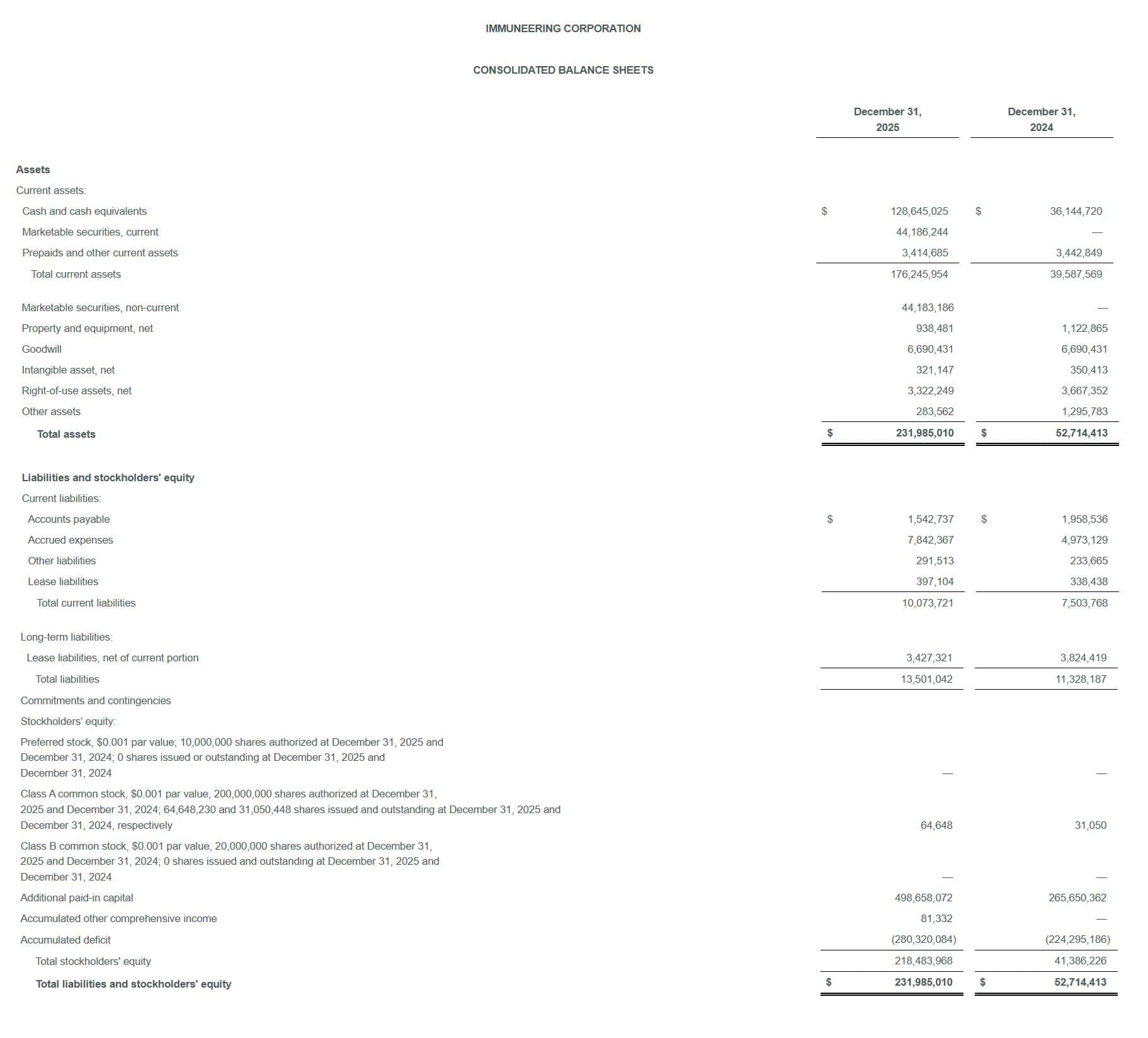
- Cash, Cash Equivalents & Marketable Securities: $217.0 million as of December 31, 2025, up dramatically from $36.1 million at year-end 2024.
- Total Assets: $232.0 million, up from $52.7 million a year earlier.
- Shares Outstanding: 64.65 million shares (Dec 2025) versus 31.05 million (Dec 2024) — more than doubled due to ~$225 million in equity financings.
- Cash Runway Guidance: Sufficient to fund operations into 2029 based on current operating plans.
- Key Clinical Milestone: 64% overall survival (OS) at 12 months in Phase 2a pancreatic cancer trial, well above the 35% standard-of-care benchmark.
- Upcoming Catalyst: First patient dosing in pivotal Phase 3 MAPKeeper 301 trial expected mid-2026.
Beat or Miss?
| Metric | Reported (Q4 2025) | Analyst Estimate | Difference/Analysis |
| EPS | ($0.18) | ($0.31) | Beat by $0.13 (~42%) |
| Revenue | $0 | $0 | In-line — clinical-stage, no product revenue |
| Net Loss | $11.6M | N/A | Improved 35.8% YoY from $18.1M in Q4 2024 |
| R&D Expenses | $9.3M | N/A | Down 37.7% YoY from $14.9M |
| Cash Position | $217.0M | N/A | Up 501% from $36.1M a year prior |
The significant EPS beat was driven primarily by a 37.7% reduction in R&D spending as the company wound down certain programs (envometinib/IMM-6-415 and pre-clinical activities), combined with higher interest income from a vastly larger cash balance following equity financings totaling ~$225 million during 2025.
What Leadership Is Saying?
“2025 was a transformative year for Immuneering. We reported 64% overall survival at 12 months in first-line pancreatic cancer patients treated with atebimetinib + mGnP, well above the benchmark for GnP standard of care. We designed atebimetinib with three mechanisms well-established to improve survival, and we believe these survival gains are driven by atebimetinib shrinking tumors durably with less resistance, preserving body mass by countering cachexia, and minimizing side effects to maximize combinability and performance status. We also made rapid progress in preparation for our pivotal Phase 3 trial in first-line pancreatic cancer patients, MAPKeeper 301, having secured alignment with both the FDA and EMA on our trial design, and we are on track to dose the first patient mid-year. With cash runway expected into 2029, uniquely encouraging clinical data, and a solid pipeline, we are strongly positioned to deliver on our mission to help patients live longer and feel better.” — Ben Zeskind, Ph.D., Chief Executive Officer
Note: The Q4 2025 earnings press release did not include a separate CFO-attributed quote on financial results. The financial commentary was incorporated within the company’s press release narrative.
Historical Performance
Q4 2025 vs. Q4 2024
| Category | Q4 2025 | Q4 2024 | Change (%) |
| Revenue | $0 | $0 | N/A |
| Net Loss | -$11.6M | -$18.1M | Improved 35.8% |
| EPS (Basic/Diluted) | ($0.18) | ($0.58) | Improved 69.0% |
| R&D Expenses | $9.3M | $14.9M | -37.7% |
| G&A Expenses | $4.5M | $3.7M | +21.6% |
| Total Operating Expenses | $13.75M | $18.56M | -25.9% |
| Interest Income | $1.86M | $0.42M | +348.3% |
| Cash & Marketable Securities | $217.0M | $36.1M | +501.1% |
| Total Assets | $232.0M | $52.7M | +340.2% |
| Shares Outstanding | 64.65M | 31.05M | +108.2% |
The dramatic improvement in net loss is attributable to reduced R&D spending on discontinued programs and substantially higher interest income from its expanded cash position. However, share count more than doubled due to the $225 million in equity financing, which diluted per-share metrics.
Competitor Comparison
Q4 2025 vs. Q4 2024
The table below compares Immuneering with Revolution Medicines (RVMD), a key competitor in the RAS-targeted oncology space, also focused on pancreatic cancer and NSCLC with its lead candidate daraxonrasib.
| Category | IMRX Q4 2025 | IMRX Q4 2024 | RVMD Q4 2025 | RVMD Q4 2024 |
| Revenue | $0 | $0 | $0 | $0 |
| Net Loss | -$11.6M | -$18.1M | -$364.9M | -$194.6M |
| EPS | -$0.18 | ($0.58) | -$1.86 | ($1.12) |
| R&D Expenses | $9.3M | $14.9M | $294.9M | $188.1M |
| G&A Expenses | $4.5M | $3.7M | $66.7M | $28.2M |
| Cash Position | $217M | $36.1M | $2.0B | ~$1.9B |
| Market Cap (approx.) | ~$322M | — | ~$20B | — |
Revolution Medicines operates at a significantly larger scale, with a cash position of $2.0 billion and a market capitalization approximately 60 times that of Immuneering. However, RVMD’s quarterly net loss of $364.9 million dwarfs Immuneering’s $11.6 million loss, reflecting its much larger clinical development footprint across multiple Phase 3 programs.
Both companies are pre-revenue clinical-stage biotechs targeting RAS-driven cancers, with pivotal pancreatic cancer readouts expected in 2026. Other notable competitors in the pancreatic cancer space include Eli Lilly (with olomorasib, a KRAS G12C inhibitor being studied in combination with atebimetinib), Regeneron (collaborating with Immuneering on atebimetinib + Libtayo in lung cancer), and BioNTech (evaluating BNT122 in PDAC).
How the Market Reacted?
Immuneering shares surged 7.03% on March 6, 2026, closing at $5.33 on a trading volume of approximately 960,000 shares, well above the stock’s recent average. The positive movement appears driven by the significant EPS beat versus consensus estimates and the company’s reaffirmed strong cash position with runway into 2029. However, shares subsequently dropped $0.28 in after-hours trading from the $5.33 close, suggesting some investors may have been taking profits or reacting to the broader clinical-stage risk profile.
The overall market sentiment remains cautiously bullish for IMRX. Wall Street analysts maintain an average 12-month price target of $15.20, implying significant upside from current levels, with targets ranging from $10.00 to $30.00 across five analysts. Mizuho Securities maintained a Buy rating on the stock as recently as March 2, 2026. The stock’s January 2026 plunge of over 23% following the 12-month survival data release — despite the data being positive — highlighted how sensitive this small-cap biotech remains to sentiment shifts and the gap between clinical results and investor expectations for Phase 2 data.




